Become a clinical trial Al organization
Verisian provides the Al infrastructure required to use Al in regulated clinical workflows

Learn how Bayer uses Verisian Al for submissions
The Information Infrastructure behind clinical trial Al

Connect your entire study
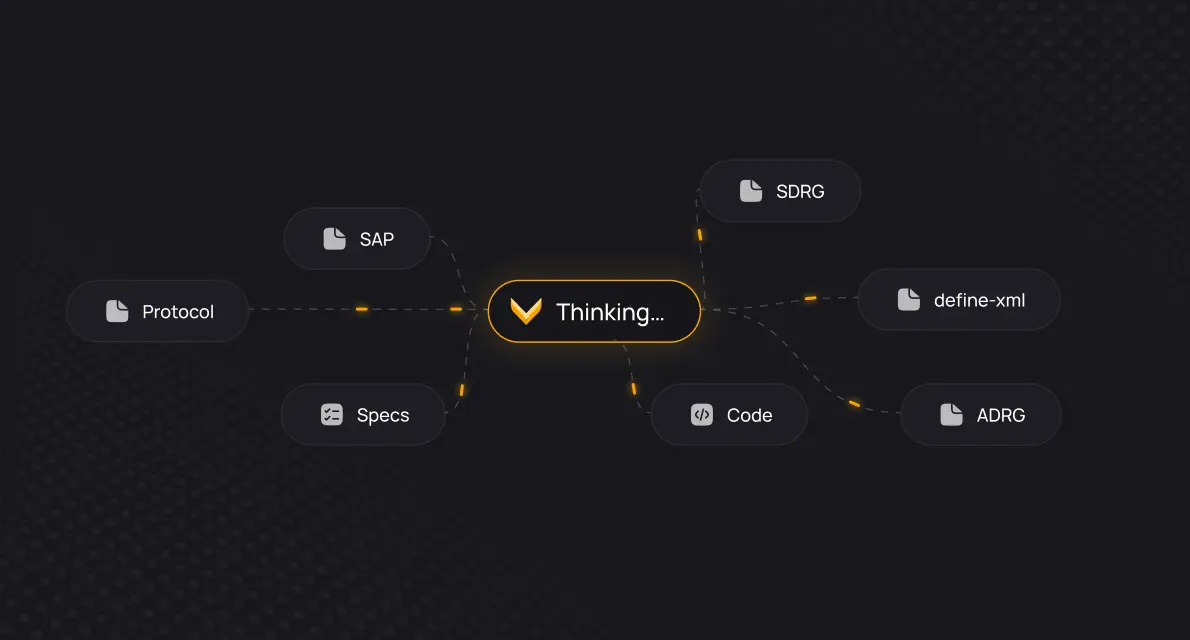
Today, clinical trial information remains deeply fragmented. Protocol, SAP, specs, code, data, results, and submission docs all exist separately and have never been truly connected.
Verisian integrates all of it through its source of truth: code. What was previously unintegratable becomes a single, machine-readable infrastructure.


Full traceability for accurate Al
Clinical trials demand perfect accuracy across hundreds of datasets, thousands of variables, and tens of thousands of dependencies. No Al can reason accurately across this volume and complexity without the right infrastructure.
Verisian connects your entire study through your code with full traceability, from specs to submission documentation. Al uses this infrastructure to retrieve exactly the right context to solve any problem as accurately as possible.

Al under expert control
Al outputs in clinical trials must be validated. Human experts need to be in control, and every result must link back to its evidence.
Every Verisian output is built on perfect context with full traceability to the code, specs, and data that produced it. Context is attached to each output, making validation simple. Human experts stay in control.

Technical Integration
Your organization, AI-enabled
Verisian integrates with any SCE or file system. No multi-year integration project. Just transfer your files and your existing infrastructure becomes AI-enabled
Connect to any SCE or file system
Your organization is AI-enabled
AI built into every process

Validation automation
AI-powered validation of SDTM and ADaM analyses

SDRG & ADRG automation
Draft reviewer's guides written by AI with guaranteed consistency

Define-XML automation
Full define-XML generation, always consistent with your data

Augmented submissions for regulators
Give regulators direct access to your study in Verisian
Submission document automation
Study planning automation
Integrated information across every stakeholder